 
From the information collected from the mass spectrometer the average mass of a carbon atom is calculated. a) 375 b) 3.75 x 10-10 c) 0.0375 d) 3.75 x 105 e) 2. detect the effect these isotopes have on the average mass. The molar mass of a substance tells you how much one mole of that substances. Carbon dioxide contains per-cent carbon and. If given the molar mass (how many g/mol of the compound) then you can calculate the. The concentration of carbon dioxide in the atmosphere is ppm. So, the molar mass of carbon dioxide is 44.01 g/mol. For carbon dioxide, the molecular formula is CO2, and the empirical. You can convert the number of moles of a substance into the number of molecules by multiplying with Avogadro’s number. The mole fraction of carbon dioxide in dry air near sea level is 3.14 x10-4 where the molar mass of carbon dioxide is 44.010. Note: You can obtain the number of moles of a substance by dividing its mass with its molecular mass. Hence, the option (B) is the correct answer. Molar mass of CO2 12.01 g/mol + (2 16.00 g/mol) Molar mass of CO2 12.01 g/mol + 32.00 g/mol Molar mass of CO2 44.01 g/mol So, the molar mass of carbon. Thus, the mass of carbon dioxide which contains the same number of molecules as are contained in 40 g of oxygen is 55 g. To determine the molecular weight (molar mass) of carbon dioxide based on measurements of the pressure, temperature, volume and mass of a sample of the gas. So, in our example, carbon has a molar mass of 12.01 grams per mole. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. Calculate the average molar mass from your three trials. So the total atomic weight of a molecule of CO2 is 44, which is 3.7 times more than the carbon atom alone weighs (44 divided by 12). The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. Molar mass of CO: Calculate the experimental molar mass of carbon dioxide for each trial using the ideal gas law. Avogadro’s number of molecules is \[6.023 \times \\ Mass of CO2: Calculate the mass of carbon dioxide in the flask for each trial from part 3 of the procedure and part 3 of the calculations. 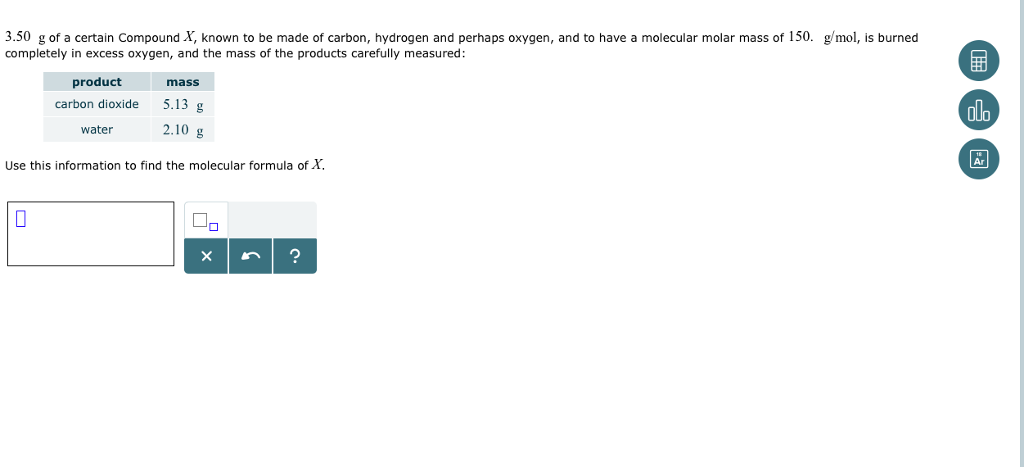
Hint: A mole of any substance contains Avogadro’s number of molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
